| :: Home :: Editor: ISSN 1715-3956 |
|
Disclaimer: |
Vol. 13 No. 1 |
April 2011 |
Commentary
Water circulation and turbulence in the cold pressor task: Unexplored sources of variance among experimental pain laboratories
Carl L. von Baeyer, David Torvi, Howard Hemingson and David Beriault
Action Editor for this manuscript was Patrick J. McGrath
In the cold pressor task (CPT), the participant places a hand or forearm in circulating cold water, which produces gradually increasing pain, and withdraws it voluntarily at any time up to a ceiling of 3 or 4 minutes (von Baeyer et al., 2005). This task can be used in experiments with children and adults to test variations in pain-altering pharmacological, attentional and social conditions. Various standardized water temperatures have been tested ranging from 0°C to 15°C, with 6°C to 10°C most common in published pediatric studies.
However, large variations in results of similar experiments performed in different laboratories might be due to an important methodological aspect of the CPT. The speed and turbulence of water circulation affect the rate of cooling of the skin and the rate of increase in pain, but have not been standardized to date. A familiar parallel, for people who live in cold climates, is the wind chill coefficient, which expresses how much colder one will feel in windy than in still conditions given the same cold outdoor temperature. Other familiar examples are the use of a fan for cooling one’s skin on a hot day, and the fact that boiled eggs or potatoes cool faster in running than in still water. With respect to cold pressor research, Mitchell et al. (2004), in a review of water temperatures used in studies with adults, confirmed that experiments using circulated water yielded lower average pain tolerance than those using uncirculated water.
The purpose of this commentary is to draw attention to this source of variability in cold pressor research findings, to discuss preliminary work on it, and to recommend that researchers consider it further in their cold pressor studies.
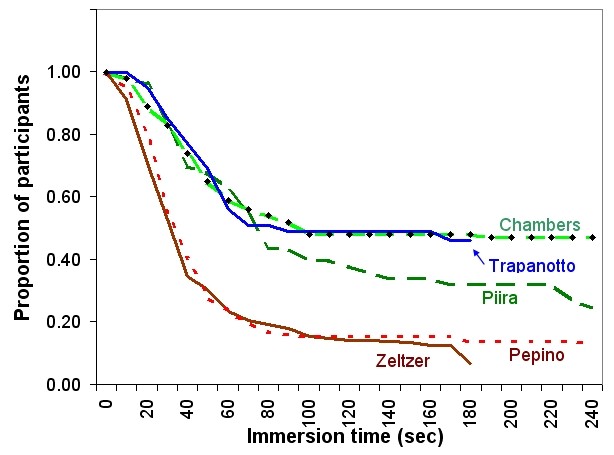
Variations in CPT apparatus for use with children are illustrated in photographs on a web page which accompanies the review article by von Baeyer et al. (2005): www.usask.ca/childpain/research/cold-pressor (accessed 1 Oct 2010). All of these laboratories used water at 10°C, yet reported markedly varied pain tolerance as shown in Figure 1.
Figure 1. Survival curves: Number of participants with their hand still in the water as a function of time elapsed. All laboratories used water at 10ºC. Trapanotto: Padua, Italy. Pepino: Philadelphia, USA. Chambers: Vancouver and Halifax, Canada. Zeltzer: Los Angeles, USA. Piira (now Jaaniste): Sydney, Australia.
The rate at which skin cools in the CPT is influenced by the rate of convection heat transfer between the cold water and the subject’s skin. Convection heat transfer is a function of water temperature, circulation in the tank, initial temperature of the skin and other factors. The rate of convection heat transfer can be estimated using the convection heat transfer coefficient. The heat transfer coefficient is expressed in SI units as watts per meter squared per kelvin: W/m2·K.
Cold pain has been studied extensively for several decades. A number of temperatures have been given for the activation of cold pain receptors in the skin, including 12°C (Nomoto et al., 2004), 15°C (Guyton & Hall, 2000), and 18°C (Wolf & Hardy, 1941). For the present commentary, it is assumed that the cold pain threshold is reached, on average, when the skin temperature reaches 15°C. (This represents a deliberate simplification, as a variety of other influences on pain threshold are present, including individual and developmental differences in pain sensitivity, properties of the layers of the skin, variations in blood circulation, and initial temperature distributions between the surface and the body’s core temperature.)
Based on a mathematical model of skin cooling, the influence of convection heat transfer on time to reach pain threshold in the CPT can be estimated, as shown in Figure 2. This model assumes initial skin temperature of 32°C, pain threshold at 15°C, and water temperature at 10°C. In this model, it takes over 50 seconds to reach pain threshold when the convective heat transfer coefficient is 500 W/m2·K, while it takes less than 8 seconds at 1,500 W/m2·K. A convection heat transfer coefficient of 500 W/m2·K would represent a still water bath, while higher values represent varying degrees of water circulation. Differences in pain threshold times predicted by the model for still and circulating water baths are similar to results reported in the literature (e.g. Pepino & Mennella, 2005; Piira et al., 2002).
Figure 2. Effect of convection heat transfer coefficient on predicted time required to reach cold pain threshold, assuming initial skin temperature of 32°C, pain threshold at skin temperature of 15°C, and water bath temperature of 10°C.

Given the influence of convection heat transfer on rate of skin cooling and hence on measures of pain experience, and disparities across laboratories in response to the CPT, it may be desirable either to measure or to control for convection heat transfer in CPT apparatus.
The simpler option would be to use standardized apparatus in all laboratories that engage in similar research. A collaboration of several CPT labs in different countries was developed starting at the Seventh International Symposium on Pediatric Pain in Sydney, Australia, in 2006, and that group agreed to recommend the use of a standard laboratory dip cooler, water bath, and thermoregulator made by Techne ® (www.techne.com, accessed 1 Oct 2010). The setup is illustrated in the top photograph at www.usask.ca/childpain/research/cold-pressor. The disadvantage of this setup is that it is expensive (in the $5,000 USD range) compared with the improvised cold pressor tanks also shown on that page. It also depends on continued availability of the exact equipment models agreed upon.
Another option would be to measure convection heat transfer, and to adjust the water circulation rate (e.g. via changing the speed or size of the circulation pump, or using baffles to direct water flow) in order to meet a specified standard value. This requires development of measurement apparatus, procedures, and normative tables or other means to analyze the results. One such project, carried out as an undergraduate engineering student project by the third and fourth authors, involved design and construction of a specialized probe for this purpose (Beriault & Hemingson, 2006). A probe was developed that is low cost (approximately $150 USD in labor and materials), relatively easy to use, and does not require the use of a sophisticated data acquisition system. It consists of a pure copper sphere, 43 mm in diameter, with a thermocouple embedded in its surface and connected by wire to a digital thermometer. The sphere is warmed to 35°C and then lowered into the cold water, and the time it takes to cool down to a specified temperature, such as 12°C, is measured in seconds. A table or spreadsheet is used to convert the measured time into a convection heat transfer coefficient.
Other possible measuring devices could be developed. Investigators have measured convective heat transfer during immersion in water by placing commercially available heat flow meters on the surface of the skin of a test subject (e.g. Castellani et al., 2007). The cost of one of these heat flow meters is in the range of $200 USD (www.conceptengineering2000.com, accessed 15 Dec 2010), in addition to the cost of the device used to record the voltage output. This measurement could be made using the arm of one of the investigators; however, placing such a device on the surface of the skin of a participant may affect CPT results. During their design project, the third and fourth authors looked at an alternative design that more closely simulates the geometry of the forearm and hand. However, this design requires a more sophisticated data acquisition system than the copper sphere or the heat flow meter.
The purpose of the present commentary was to draw attention to variations in convection heat transfer as a potential source of discrepancies in CPT research results across laboratories. Within labs, as long as the same apparatus and flow conditions are used, this is not an issue, but difficulties in reconciling conflicting findings across laboratories may be reduced by use of standardized equipment and protocols, or by measurement and calibration of equipment to meet an agreed upon standard of convection heat transfer in the CPT. The influence of water flow rate and turbulence on convective heat transfer is, of course, only one potential source of variation across laboratories. Others might include ambient temperatures, cultural or ethnic differences, procedural differences, or children’s typical experience with cold water, each of which might deserve separate investigation.
Carl L. von Baeyer, PhD David Torvi, PhD, PEng Howard Hemingson, BSc, BE David Beriault, BE |
Cite as: von Baeyer CL, Torvi D, Hemingson H, Beriault D. Water circulation and turbulence in the cold pressor task: Unexplored sources of variance among experimental pain laboratories. Pediatric Pain Letter 2011;13:13-16. ppl.childpain.org
References
Beriault D, Hemingson H. Measurement of the convective heat transfer coefficient in a cold pressor apparatus. ME 495 Design Report, Department of Mechanical Engineering, University of Saskatchewan, Saskatoon, SK, 2006.
Castellani JW, O’Brien C, Tikuisis P, Sils IV, Xu X. Evaluation of two cold thermoregulatory models for prediction of core temperature during exercise in cold water. J Appl Physiol 2007;103:2034-2041. PubMed Abstract
Guyton AC, Hall JE. Textbook of medical physiology (10th ed.). Philadelphia: WB Saunders, 2000. p. 562. Link
Mitchell LA, MacDonald, RAR, Brodie EE. Temperature and the cold pressor test. J Pain 2004;5:233-237. PubMed Abstract
Nomoto S, Shibata M, Iriki M, Riedel W. Role of afferent pathways of heat and cold in body temperature regulation. Int J Biometeorol 2004;49:67-85. PubMed Abstract
Pepino MY, Mennella JA. Sucrose-induced analgesia is related to sweet preferences in children but not adults. Pain 2005;119:210-218. PubMed Abstract
Piira T, Taplin JE, Goodenough B, von Baeyer CL. Cognitive-behavioural predictors of children’s tolerance of laboratory-induced pain: implications for clinical assessment and future directions. Behav Res Ther 2002;40:571-584. PubMed Abstract
von Baeyer CL, Piira T, Chambers CT, Trapanotto M, Zeltzer LK. Guidelines for the cold pressor task as an experimental pain stimulus for use with children. J Pain 2005;6:218-227. PubMed Abstract
Wolf S, Hardy JD. Studies on pain. Observations on pain due to local cooling and on factors involved in the “cold pressor” effect. J Clin Invest 1941;20:521-533. PubMed Abstract

